Equation maker chemistry2/19/2023  
Which means that all coefficients depend on a parameter a, just choose a=1 (a number that will make all of them small whole numbers), which gives: Solve the system (Direct substitution is usually the best way.) So, for each element, count its atoms and let both sides be equal.ģ. There must be the same quantities of each atom on each side of the equation. a K 4Fe(CN) 6 + b H 2SO 4 + c H 2O = d K 2SO 4 + e FeSO 4 + f (NH 4) 2SO 4 + g COĢ.(Coefficients represent both the basic unit and mole ratios in balanced equations.): In reactions involving many compounds, equations may then be balanced using an algebraic method, based on solving set of linear equations.ġ. Since there are 7 oxygen atoms on the right and only 3 on the left, a 3 is placed before O 2, to produce the balanced equation: C 2H 5OH + 3O 2 = 2CO 2 + 3H 2O Using Linear Systems 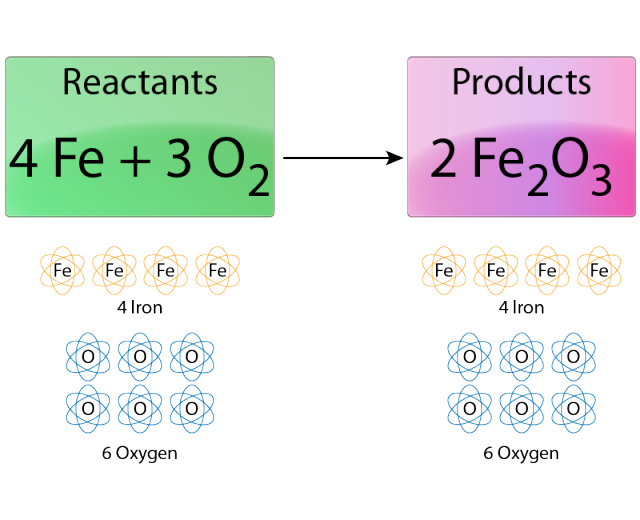
Since C 2H 5OH contains 6 hydrogen atoms, the hydrogen atoms can be balanced by placing 3 before the H 2O: C 2H 5OH + O 2 = 2CO 2 + 3H 2Oįinally the oxygen atoms must be balanced. The most complicated molecule here is C 2H 5OH, so balancing begins by placing the coefficient 2 before the CO 2 to balance the carbon atoms. This equation is more complex than the previous examples and requires more steps. Example #3 (Complex) C 2H 5OH + O 2 = CO 2 + H 2O The equation is now balanced because there is an equal amount of substances on the left and the right hand side of the equation. To fix this unbalanced equation a 5 in front of the O 2 on the left hand side is added to make 10 O's on both sides resulting in P 4 + 5O 2 = 2P 2O 5 The left hand side has 2 O's and the right hand side has 10 O's. The left hand side has 4 P's and the right hand side has 4 P's. 
This equation is not balanced because there is an unequal amount of O's on both sides of the equation. This equation is a balanced equation because there is an equal number of atoms of each element on the left and right hand sides of the equation. 
The equation will now look like this: 4Na + O 2 = 2Na 2O To fix this 2 more Na's are added on the left side. Again, this is a problem, there must be an equal amount of each chemical on both sides. The right hand side has 4 Na's total and 2 O's. Currently the left hand side of the equation has 2 Na atoms and 2 O atoms. Notice that the 2 on the right hand side is "distributed" to both the Na 2 and the O. Now the equation reads: 2Na + O 2 = 2Na 2O To fix this a 2 is added in front of the Na 2O on the right hand side. On the left hand side there are 2 O atoms and the right hand side only has one. In the next step the oxygen atoms are balanced as well. In this there are 2 Na atoms on the left and 2 Na atoms on the right. This problem is solved by putting a 2 in front of the Na on the left hand side: 2Na + O 2 = Na 2O As it stands now, there is 1 Na on the left but 2 Na's on the right. In order for this equation to be balanced, there must be an equal amount of Na on the left hand side as on the right hand side. Hydrogen and oxygen are usually balanced last. Generally, it is best to balance the most complicated molecule first. Simple chemical equations can be balanced by inspection, that is, by trial and error. Using Trial and Error/Inspection Example #1 (Simple) By changing the scalar number for each molecular formula, the equation may be balanced. In case of net ionic reactions, the same charge must be present on both sides of the unbalanced equation. Thus, each side of the equation must represent the same quantity of any particular element. In a chemical reaction, the quantity of each element does not change. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
